|
Air pollution from industrial production

|
The title of William S. Johnson's autobiography,
A Fifty-Year Love Affair with Organic Chemistry,
could not be more fitting. Indeed, Johnson did have a 50-year love
affair with organic chemistry, and his contributions to the field were
enormous. In an epilogue, Paul A. Bartlett, William R. Bartlett, John D.
Roberts, and Gilbert Stork wrote: "Bill Johnson's work didn't just
alter the way we approach synthesis, it helped change our view of what
is even possible. As a result, an entire continent, stereospecific
cationic cyclization, has been added to the world of organic chemistry.
When he began his historic work on the polyene cyclization problem, no
one seriously imagined that a complex structure like that of a natural
steroid could, one day, be assembled with essentially complete regio-
and stereospecificity; some actually stated so quite strongly! Although
we are amused at how stunningly wrong this view turned out to be, we
must not overlook the fact that when Johnson started his career, the
concept of controlling the stereochemical course of a sequence of
organic reactions was unknown. The few syntheses of natural products
that had been recorded only served to emphasize the improbability of
success in assembling more complex structures."
I want to discuss a topic, climate change, which I think will
affect our lives and our science dramatically in the coming years.
As Bill McKibben observes in his recent book
Eaarth: Making a Life on a Tough New Planet, many politicians and other policy
makers have had a habit of discussing global warming as a phenomenon
that will affect future generations unless we do something now to avert
it. However, that's just not the case. Climate change is occurring right
now. It's not a problem for our children and grandchildren; it's a
problem for us.
In fact, the terms climate change and global warming
may be misleading. I attended the Kavli Award ceremonies in Oslo in
September. At a policy forum on international collaboration in science
and technology, President Barack Obama's science adviser, John Holdren,
made an interesting point. He said: "Global warming is a dangerous
misnomer. It suggests that the changes are uniform, primarily about
temperature, gradual, and likely benign. None of these are true." What
we should be calling the phenomenon, he said, is "global climate
disruption."
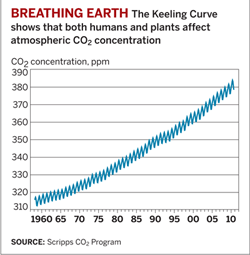
I'm sure many of you are familiar with
the plot of the concentration of atmospheric CO2
at 3,000 meters on Mauna Loa in Hawaii over time. It's called the
"Keeling Curve" after Charles David Keeling, the man who made most of
the measurements. Keeling received his Ph.D. in geochemistry from
Northwestern University. He began measuring CO2 as a postdoc
at the California Institute of Technology in the mid-1950s in places
like Pasadena and Big Sur. He took a position at Scripps Institution of
Oceanography in 1956 and started measuring CO2 on Mauna Loa in 1958.
This is a truly remarkable data set. Of course, it shows that atmospheric CO2
concentrations are rising, but that really should not have been
surprising. No one had measured it before, but given the amount of
fossil fuel that was being burned, a simple mass-balance analysis
guarantees the observed increase over time. The only thing in doubt is
the slope of this line because we didn't know then, and we still don't
have a very good understanding of, the role of the oceans in absorbing
CO2 from the atmosphere.
What's truly remarkable about this plot is that it shows
that Earth breathes, one breath per year. Each decrease in CO2
is an inhalation, each increase an exhalation. Keeling understood what
he was seeing immediately. Because the majority of Earth's landmass is
in the Northern Hemisphere, there are more plants in the Northern
Hemisphere than in the Southern Hemisphere. In the spring and summer,
those plants are absorbing CO2 as they grow and produce
leaves. In the fall and winter, the leaves fall and decompose.
One of the most unnerving aspects of global climate
change for the human psyche to absorb is that it drives home with
absolute finality the notion that Earth is finite. I know, that sounds
obvious, but people have never behaved as if Earth were finite. They
have behaved as if Earth and its resources, the environment itself, were
infinite.
The Keeling Curve doesn't demonstrate that climate is
changing; it simply provides the evidence that supports the most obvious
mechanism to account for the global warming that has also been
measured. It does show, in two very concrete ways, that Earth's
atmosphere is finite and can be impacted by the biosphere. Humans
account for the increase over time; plants account for the annual
periodicity.
 |
If Earth is finite, then by definition, so is our
capacity to produce and consume. Yet we exist within a socioeconomic
system that is predicated on endless growth. The rate of growthin
population and economic activityturned exponential about 200 years ago
with the advent of the Industrial Revolution. We call it the Industrial
Revolution, but that revolution was really a revolution based on the
extraction of fossil fuels from Earth and their use to power machines
and eventually to produce electricity. Civilization as we know it is
entirely dependent on burning fossilized sunshine cheaply. Because
that's what fossil fuels areyes, they're the fossils of dead plants and
dinosaurs, but those flora and fauna were just the machines that
converted hundreds of millions of years of sunshine into compounds
buried in Earth waiting for us to extract them. We burned the fossil
fuels on the cheap because we treated the atmosphere as a free dumping
ground for the waste products of combustion, primarily CO2.
Eventually, humanity was going to hit a wall. A wall
that told us that a system based on endless growth was not sustainable
on a finite planet. What seemed obvious was that, at some point, we were
going to use up Earth's reserves of fossil fuels. In the case of
petroleum, some experts believe we have already reached or will very
soon reach "peak oil," the point at which we have consumed half of all
the petroleum on Earth and at which point production will begin to
decline. Whether we've reached peak oil or not, we've certainly reached
the peak of oil that's reasonably easy to extract from Earth. Otherwise,
why are we drilling in 5,000 feet of water through another 13,000 feet
of rock in the Gulf of Mexico? Why are we contemplating drilling in the
Arctic Ocean or in 2 miles of water off the coast of Brazil?
It turns out the availability of
fossil fuels wasn't the wall that put a limit on growth; climate change,
global warming, climate disruptionwhatever you want to call itturned
out to be the wall. There are enough fossil-fuel resources left on Earth
for us to keep the economic engines that have powered 200 years of
exponential growth going for another 100 or 200 years or so, but the
climate isn't going to let us do that.
An aside, though. Yes, enough fossil-fuel resources
exist for us to continue in a business-as-usual mode for some time, but
at what cost, even leaving climate change aside? We have no idea of the
long-term environmental damage caused by the BP oil spill in the Gulf of
Mexico. We're blasting the tops off of mountains in West Virginia to
get at the coal beneath them, in the process destroying hundreds of
miles of pristine streams and despoiling hundreds of square miles of
landscape, essentially forever. How much of our environment are we
willing to degrade to satisfy our addiction to fossil fuels?
|
Surface coal mining in Wyoming, USA

|
Oil drilling off California coast, USA

|
The fact is that, eventually, we have to learn to live
off the sun in real time. That's not going to be easy. Fossil fuels
aren't just fossilized sunshine; they're concentrated fossilized
sunshine. As McKibben points out in "Eaarth," 1 barrel of oil yields as
much energy as 25,000 hours of human manual labormore than a decade of
human labor per barrel. The average American uses 25 bbl per year (some
estimates are quite a bit higher), which, he writes, is like finding 300
years of free labor annually.
To live off the sun in real time, we're going to have
to do two things: We're going to have to slow down, and we're going to
have to get a lot smarter. Slowing down will involve making the
wrenching transition to an economic system that is not predicated on
growth. I don't know what that system looks like. In my mind, I have a
notion of something I call a high-tech subsistence economy in which
consumption is not the sine qua non of success.
And we're obviously not going to make the transition to
living off the sun in real time in a year or two or even a decade or
two. It will require a transition period during which we still burn
fossil fuels, but hopefully learn to burn them in a cleaner fashion.
During which time we build more nuclear power plants to produce
electricity. During which time we adapt to a changing, disrupted
climate.
As Holdren pointed out in his talk in Oslo, humanity
has three options in the face of climate change: mitigation, adaptation,
and suffering. "We're already doing some of each," he said. What's up
for grabs is the ultimate mix of the three. Mitigation can't work alone
because climate change is already happening and will continue regardless
of what changes humans make in their use of energy. We will have to
adapt to an altered climate. We're already adapting, whether we
acknowledge it or not. The new flood walls circling New Orleans are an
adaptation to climate change. And unless humans are able to limit global
warming to no more than 2 °C on averagewhich at this point is very
much in questionthe suffering humanity faces is going to be severe.
What role will chemistry and the chemistry enterprise
play in this? We don't call it the "central science" without good
reason, and in this world of a changing, disrupted climate and an
evolving economic system, chemistry will take on an even more important
role than it has in today's society and economy.
Green chemistry and green engineering will obviously
play an important role in creating more sustainable manufacturing
practices. They're already having an impact. Earlier this year, I
interviewed Paul T. Anastas,
the assistant administrator in charge of the Environmental Protection
Agency's Office of Research & Development and one of the fathers of
the green chemistry movement. He said to me, "My vision for the future
of the Office of Research & Development includes a recognition that
the goal of sustainability is our true north,' that scientific and
technological innovation is essential to the success of our mission,
that we need to couple our excellence in problem assessment with an
equal excellence in solving problems, and that we must act with a sense
of urgency."

NASA
NORTHWEST PASSAGE Image of Arctic
ice conditions at the end of the melt season in 2007 shows large areas
of open water and an ice-free opening through the Northwest Passage that
lasted for several weeks.
|
In our interview, however, Anastas also referred to a
speech EPA Administrator Lisa P. Jackson had given in early March in
which she said, "It's time to put to rest the notion that economic
growth and environmental protection are incompatible. It's time to
finally dismiss this false choice." That's a comforting notion, but it
is one that is no longer true. Sustainable economic growth is an
oxymoron.
Another chemical concept that will be
key to our future is "atom economy," a concept originally developed by
Stanford University chemistry professor Barry M. Trost in a seminal paper in Science (1991, 254,
1471). Certainly the atom economy of chemical reactions is key, but I'm
talking about atom economy more broadly defined, not just in chemical
reactions but in the life-cycle analysis of all products. Where do the
atoms that go into a product come from, and where do they go at the end
of the life of the product? We're going to have to get much, much better
at accounting for and conserving all of those atoms if we are going to
develop truly sustainable manufacturing processes.
Of course, neither green chemistry and engineering nor
atom economy directly addresses the energy challenge that is at the
heart of the climate-change dilemma. Learning to live off the sun in
real time is a challenge that must be addressed by chemistry and the
chemical industry. We're doing it now, but not with nearly the sense of
urgency that's required. We need to develop over the next few decades
mechanisms for the large-scale conversion of sunlight into the
electricity and liquid fuels needed to power civilization. We need
batteries to store massive amounts of electrical energy generated by
solar and wind power.
These are all challenges that fall under the category
of mitigation of climate change. They are mechanisms to head off the
worst-case scenarios we can imagine. Chemistry and related disciplines
will also be called on to contribute to the inevitable adaptations
humans will have to adopt in the face of a changing climate. We will
need crops that tolerate higher temperatures and can survive on less
water. We will need new herbicides and insecticides to deal with pests
that migrate with the changing climate. We will need new
pharmaceuticals. Chemistry, in fact, is the fundamental science that
will be called on to preserve any semblance of the quality of life to
which we have become accustomed if the worst-case climate-change
scenarios come to pass.
Are we up to the task? Certainly, chemists around the
world are making important contributions on all of these fronts, as
C&EN reports each week. When I visit chemistry departments, I am
continually impressed with the vigor and enthusiasm on display. I saw it
today in numerous posters on work being done here at Stanford. I have
talked for many years about chemistry both as a core discipline and as
an enabling science. Both aspects of our science are important. It is as
an enabling science, however, that chemistry will address many of the
challenges posed by climate change. I see chemists increasingly working
in multidisciplinary teams with biologists, physicists, materials
scientists, physicians, engineers, and others in focused research that
is leading to new drugs, new materials, new catalysts, and new processes
to meet the ultimate goal of a sustainable economy.
On the other hand, climate scientists such as James E.
Hansen of Columbia University and Stephen H. Schneider of Stanford have
been warning us about the growing threat of global warming for at least
25 years, and we've done almost nothing to change the disastrous course
we are on. Chemical scientists are optimistic about what they can invent
and create to help with the mitigation of and adaptation to climate
change as well as the creation of a sustainable economic system, but not
nearly enough is being done. There is no political will to solve this
problem because solving it requires sacrifice, and almost no one,
especially here in the U.S., is willing to ask humans to sacrifice.
In the Sept. 13 issue of C&EN, there were two News of the Week stories on one spread. On one page, there was a story with the headline Reducing Carbon from Coal, which discusses two initiatives in Germany for capturing CO2 from coal-fired power plants and using it as a chemical feedstock. On the opposite page was a story with the headline A Bleak View for Curbing CO2, which reports on a study in Science magazine that shows that it will be next to impossible to reduce CO2
emissions in a meaningful way over the next 50 years. So it's actually
pretty likely that humans are going to create a world in which CO2
concentrations double from the preindustrial level of about 280 ppm to
500 to 600 ppm, resulting in an average increase in global temperature
of at least 4 °C. In that case, it will be nature that imposes the
transition to an economy that is not based on growth. It will not be
humans making a conscious choice to develop such an economy. It will be a
difficult transition.
"World Made by Hand" is a novel by James Howard
Kunstler that I learned of from reading McKibben's "Eaarth." It's a
dystopian fable of the world some number of years from now, a world with
a drastically changed climate and a complete breakdown in the social
order. It is a world in which the machine age has ended, and the major
forms of energy are human and animal muscle power. It is, literally, a
world made by hand. Is this a likely scenario for our future? Probably
not. But it's also not outside of the realm of the possible. James
Lovelock, originator of the Gaia Hypothesis, is far more pessimistic
about the impact of global warming on humanity's future than Kunstler
is. And Jared M. Diamond's book "Collapse" shows clearly how complex
societies can implode quite rapidly, especially in the face of rapid
environmental changes.
I'm not as pessimistic about humanity's future as
Kunstler is. Humans are amazingly adaptable and amazingly creative. But
cheap energy in the form of fossil fuels burned without any concern for
their impact on climate has allowed humans to be stupid in the way we've
developed our civilization. It's as simple as that. We have no choice
but to get smarter. And we have to do it very quickly. I think it is up
to scientists to lead this charge, and lead it in two ways. One is
through research to mitigate the worst aspects of climate change and
help humans adapt to the change that is inevitable. But at least as
important, we must also be vocal advocates for changing the course we
are on. Why? Because not enough people with the credibility of
scientists have taken up this cause with the passion it deserves. We're
talking about the future of Earth. Because scientists know that this
problem is real and serious and can explain that the arguments of those
who deny that it is real and serious are specious.
You know, we haven't always been known
as scientists. I learned one day driving home from work listening to
National Public Radio's "Science Friday" that "scientist" is a word with
a definite origin. It was coined in 1834 by Cambridge University
historian and philosopher of science William Whewell. The older term for
people pursuing scientific activities, "natural philosopher," was in
Whewell's view "too wide and too lofty."
Perhaps natural philosopher is too wide and too lofty a
term to describe people who do science. However, I think we may have
given something up when we abandoned natural philosopher for scientist.
It isolated us, allowed us retreat into pursuing scientific discovery
for its own sake, and in doing so, cut science off from the humanities
and scientists off from their responsibilities as citizens. I don't know
whether anything we do can change the course we're on, but I know we
have to try. If scientists are the ones who know that global climate
disruption will be a human tragedy of as yet undefined proportions and
we do nothing about it, what does that say about us? Why should society
support us? What is our role as scientist citizens?
I am not saying that we should go back to calling
ourselves natural philosophers. I am saying that scientists, especially
chemists, who are among the most practical of scientists, must become
far more involved in this most important policy debate of our time.
Views expressed are those of the author and not necessarily those of ACS
Copyright © 2010 American Chemical Society